Currently there are different techniques to plate, and each one of them has a specific use and properties. Some also require prior procedures such as serial dilution to be performed. So, although we have different ways of plating or monitoring microbial growth in culture, four of them are the most used: the spread plate, the pour plate, the drop plate, and the spiral plate.
IUL Instruments designs, manufactures, and commercializes classical microbiology devices and consumables, such as the methods that are going to be mentioned in this post. With the ultimate technology, IUL offers solutions for laboratories in sample preparation, serial dilution, plate inoculation, and colony counting, and more.
Spread Plate
The spread plate technique involves using a sterile L-shape glass rod (Spreader) with a smooth surface made of metal or glass to apply a small number of bacteria suspended in a solution over a Petri dish so that the bacteria are easy to count and isolate.
According to FDA BAM (Chapter 3), the suitable colony counting range is 25-250 colonies per plate. Plates with more than 250 CFUs are considered too numerous to count or may inhibit the growth of some bacteria. On the other hand, plates with less than 25 colonies do not have enough colonies to consider the plate statistically representative of the analyzed sample.
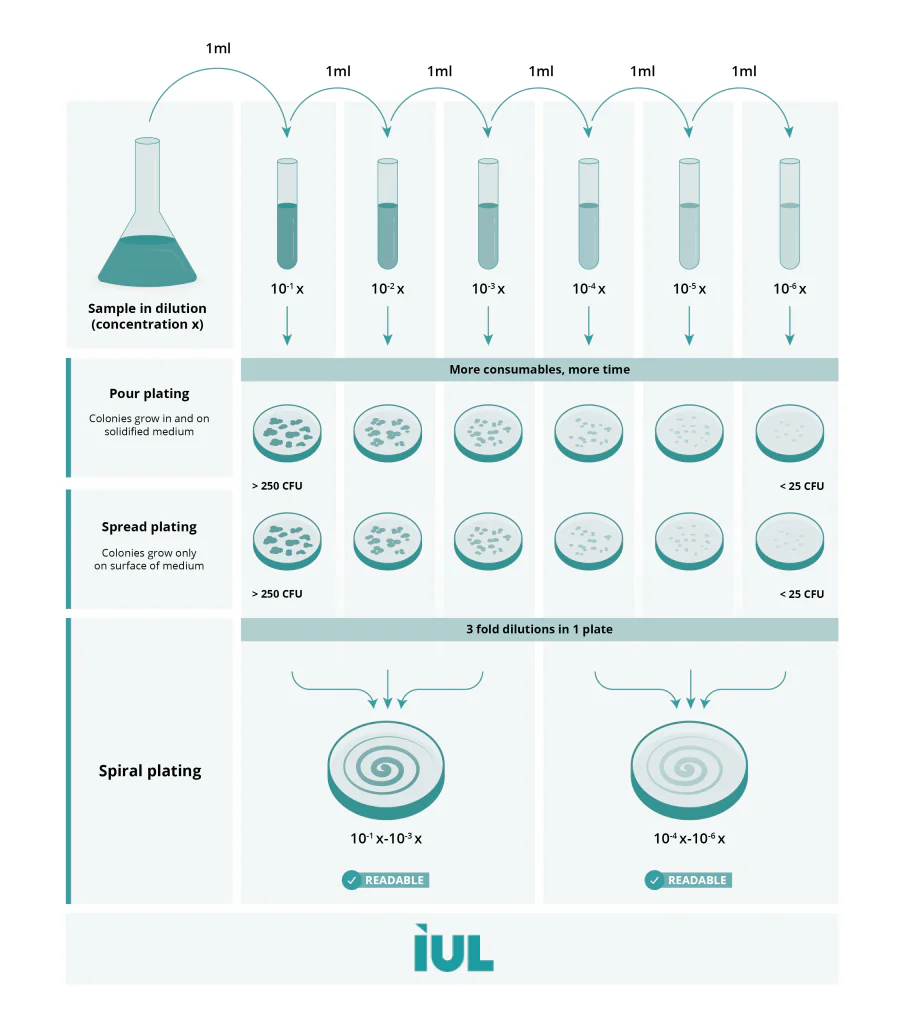
Therefore, to get optimum results from spread plating, before inoculating the sample on the plate we need to perform a serial dilution and plate at least three plates. With this procedure, the sample is diluted through a series of standard volumes of sterile diluent. By diluting the sample, it is possible to plate different concentrations of a sample and obtain an incubated plate with an easily countable number of colonies and calculate the number of microbes present in the sample.
Pour Plate
The pour plate is a technique of choice for counting the number of colony-forming bacteria present in a liquid specimen. With this method, an amount of bacteria suspended in a solution (around 1 ml) is placed in the center of a sterile Petri dish using a sterile pipette. Molten cooled agar is then poured into the Petri dish containing the inoculum and mixed well. After the solidification of the agar, the plate is inverted and incubated as usual.
This technique also requires a prepared sample like the spread plate, performing serial dilutions of the mixed cultures to obtain the optimum number of colonies present in a plate to perform the count.
Drop Plate
The drop plate method can be used to determine the number of viable suspended bacteria in a known beaker volume. The drop plate exhibits many positive characteristics. The plating and counting procedures require less labor than alternative methods, and the plating and counting steps are very convenient and manageable. Also, less time and effort are required to dispense the drops onto an agar plate than to spread an equivalent total sample volume into the agar.
This method allows dispensing the sample to be analyzed in drops, colony counting can be done faster and perhaps more accurately. Furthermore, the drops can absorb quickly into the agar on appropriately dried plates. Besides this, the drop plate method expends relatively few supplies but, despite its widespread use, the drop plate method has not stardardized yet.
Spiral Plate
Spiral Plating is a two-in-one method: dilutor and plater at the same time, one of the most time and waste volume saving methods of inoculation, which means cost savings, that is why more and more microbiological laboratories are using a Spiral Plater for baterial determination.
This rapid plating consists of inoculating a known volume of a liquid sample on a surface of a rotating agar plate. The volume of the sample that is dispensed, decreases as the dispensing syringe moves from the center to the edge of the rotating plate doing an Archimedean spiral pattern. As a result, versus plating manually, different concentrations of the sample are present in one plate and the time spent on colony counting is shortened. No serial dilutions are required!