Abstract
Indeed, food quality control and safety is a complex topic and many different ideas and point of views are present. The tricky thing is to find exactly the methods and instruments which provide the results needed for good decision if a batch is fine or not. Of course, expertise for interpretation of those analytical values is also important to profit from the different analyses.
One of the most neglected parameter in the food quality control world is water activity. Normally, it is used for microbial risk determination and mitigation but limits are often that far beyond actual values, that no one really cares, if water activity varies a bit; as long as it stays within the limits, all is fine. But at the same time, the opportunity for production optimization is missed and money is still going to waste by production problems, non-conformity of texture, taste etc. No worries, help is just around the corner: It’s water activity measurement..
Introduction
Before talking seriously about water activity, one must know what water activity (aw) is.
Water activity has its roots in the classical thermodynamics. It is defined as the vapour pressure over a sample divided by vapour pressure over pure water, both at the same temperature:
aw = vapour pressure over sample / vapour pressure over pure water
In fact, it is an activity (or potential) in the physical-chemical sense and has just little to do with the amount of free water in a product. It is all about interaction of the free water with the product. Reference is pure water which has the highest possible activity. The more interaction there is with the product, the less “active” the free water becomes and thereby water activity goes down.
In addition, water activity is not the same as moisture. Moisture describes the total water in a product. And here is the problem. If you have 5% moisture, how much of that total water is free and how much is bound? Without this important information, nothing can be said about microbial risks, texture changes and so on.
To determine the aw-value, relative air humidity over a sample is measured after reaching equilibrium between free water in and air humidity in the air above the sample. The detected air humidity is divided by 100 to get the aw-value.
Aw = ERH [%] / 100
Aw: Water activity value
ERH: Equilibrium Relative (Air) Humidity
An accurate, repeatable and reliable aw-measurement is only possible, if the sample has constant temperature during measurement and if an instrument takes the measurement once humidity equilibrium has been established. Thus a temperature controlled measuring chamber and automatic equilibrium detection is absolutely mandatory.
Where can you profit from aw?
The water activity value itself does not say anything; you need additional info (tables, tests etc) to link it with an effect like bacterial growth, oxidation or others.
Most of the clients are using water activity measurement day by day for microbial risk determination. They have aw limits and just make sure to stay below them. Most of the limits are set in a way to avoid growth of bacteria, especially the pathogenic ones but by just focusing on this, chances are forgone to optimize a product in chemical and physical matters and thus save money in production.
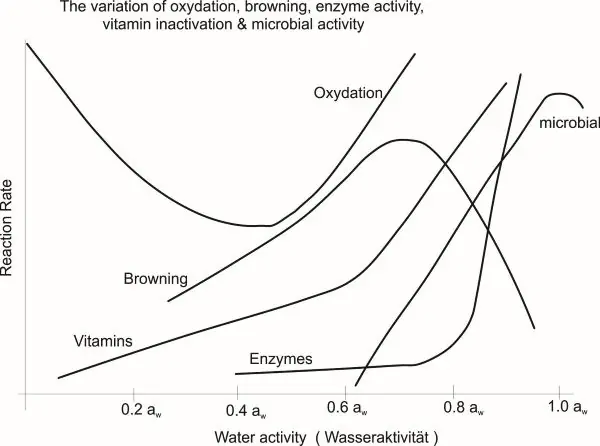
[1] Food Stability Map as a function of water activity
Let’s look a bit behind the scene of the most common industries where water activity is used and see those opportunities for optimization.
Powder industry (milk powder, spices, ingredients…)
Even if measurement of water activity in powders seems to be an easy deal, interpretation can become quite tricky.
Even more so that sudden product texture changes, discoloration, oxidation, caking & clumping or hardening cannot be further explained. Water activity value and all other parameters are within specs.
Looking a bit closer to what is happening on material level. To create powders, drying steps are crucial but they are also the source of problems. During this process, doesn’t matter which technology (spray or drum drying or even lyophilization) is used, product size is shrunk (drops to particles). This will create capillaries in which free water is held back due to capillary forces. As drying happens at the outer surface of a particle, the inner side contains more water than the outside, so there is a risk that inner water activity is not equal to surface water activity

But what are people doing if product’s aw has to be measured? Correct, they take samples right after the drying process or once production has been finished. If water activity is measured at that point, you will get the surface aw of the particle but no chance to get the water from the inner side as it takes much longer till thermo-dynamical equilibrium of water in those particles has been reached.
There are a few approaches published by use of sorption isotherms to solve this problem.
A new possible solution came up by using thermal treatment of the powder. This is rather usable for single ingredients with glass transition temperature (Tg) in the range of 25-60°C.
It is important to know that Tg depends on aw-value of the sample, respectively on air humidity of the surrounding air.
The idea is to heat the ingredient above its glass transition temperature for hours to break the capillaries and all internal structures; afterwards cool down. This way, the free water in the capillaries becomes detectable as nothing retains it. This will lead to higher but real water activity values once product is completely in thermo-dynamical equilibrium.
All this has to be done preferably in the water activity meter itself to avoid loss of free water. So, perfect chamber sealing is more than crucial.
Meat and Fish
Economy of product, these are the magic words for that kind of industry. If water activity regulations exist in a country, most probably they do for meat products,. Specifically for Listeria, there is a clear 0.92aw limit. Every producer’s goal is to implement as much water as possible in the product but without the risk of Listeria spoilage. Every kilogram of water which can be left in the product lowers the material costs and enhances the profit. In terms of that fact, microbial safety is the main focus.
But if products are dried like jerky style meat or fish, non-enzymatic reactions become very important. Especially the so called browning, which is a recombination of proteins and reducing sugars makes a product bitter in taste and thereby no more enjoyable. Bringing aw-values down to 0.2aw would help but process-wise not really possible, adjusting drying temperature and time is the key to avoid it. But why is this a water activity topic? Well, free water transports all reactants to the point of action. No free water, no non-enzymatic reaction.
Bakery, Sweets, Chocolate..
Beside microbial safety, this industry deals with water migration between multi-layers (like waffle-chocolate sandwich, glazing and batter, chocolate chunks in cookies,..) and fat oxidation. While fat oxidation can be reduced by processing and storing the goods with an adjusted aw-value around 0.45aw – 0.50aw, water migration is a challenge. In principle, free water moves between areas of different water activities and not between different moisture contents. To get rid of water migration issues, all layers in a product must have the same water activities but here comes the challenge. Lowering or increasing water activity goes together with sensory/texture/colour changes. You cannot ad 1kg of salt to 10kg of product.
It is a game at the edge of all possibilities and finally a compromise. But understanding how the water migrates will create a more stable products and less risk of chemical/physical changes in it.
Pharma
It is possible to reduce waste during coating process if water activity of the tablet is within specified limits or it is possible to determine the hygroscopicity of an API or formulation to optimize the production process (exposing time to air) plus finding best storage conditions and packaging.
Water migration is a second field of interest, especially if soft gel capsules are involved. It is of tremendous interest to know how free water migrates as the shell might dry out and become too hard (and get cracks) or get wet and loose its physical properties. Optimization of the filling can prevent such issues but here as well, changing the filling water activity, solubility, texture and maybe stability is changing as well.
An easy combination of water activity information with water content using sorption isotherms opens much more details about water kinetics and free water behavior in a pharmaceutical product.
Cosmetics
Cosmetics is a special discipline in the water activity measurement business. Problem here is that most of the products are emulsions, stuffed with talc, glycerine (softeners), fragrances etc., so a fierce mix of various chemicals as well as natural products. For emulsion or other liquid cosmetics, water activity can be used to determine the risk of microbial spoilage. Another aspect which cannot be neglected is the separation of water and oil/fat plus the oxidation of fat which spoils the cosmetic product (gloss, bald, cream / lotion,..). It is important to manage the aw-value within tough limits to avoid such unfortunate incidences.
In addition, water activity fits perfectly in a HACCP concept and is used as a control parameter at several production steps so that short time intervention can be made once the parameter is not within the limits.
References:
[1] Food Stability Map as a function of water activity (adapted from Labuza, T.P., Tannenbaum, S.R. and Karel, M. 1970, Journal of Food Technology 24:543-550)